https://www.nature.com/articles/s41467-024-45545-z
https://www.cas.cn/syky/202402/t20240218_5005493.shtml
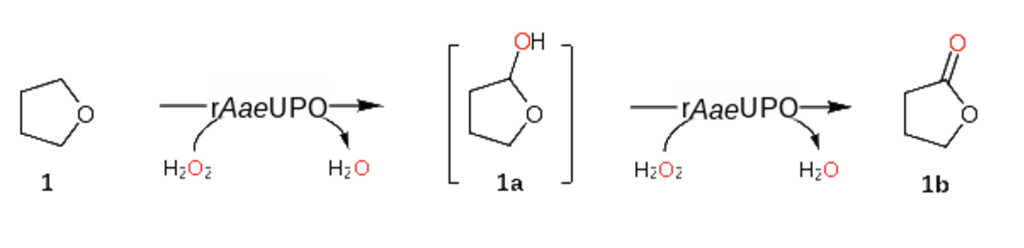
A team at the CAS Tianjin Institute of Industrial Biotechnology has applied reaction engineering strategies using immobilized peroxygenase from Agrocybe aegerita (AaeUPO) for C-H bond selective hydroxylation reaction of cyclic ether molecules in organic solvents, for the synthesis of optically pure chiral hemiacetal compounds .
The immobilized peroxygenase which was already known to convert tetrahydrofuran into hemiacetal products also transformed tetrahydropyran rings to hemiacetals, with a selectivity of 97%, a TON of up to 95172, and an enantiomer selectivity (ee) of over 99%. For other series of products with chiral carbon atoms, the ee value of the corresponding hemiacetal product can exceed 99%.
Using reaction engineering to further study how the hemiacetal product obtained by hydroxylation of C-H bonds in the aqueous phase could be further oxidized in situ to obtain lactone compounds in a one-step method, it was showed that the ratio of hemiacetal and lactone products differed at different pH. At pH 4.0, only the hemiacetal product was detected, at pH 9.0 only the lactone. Based on this discovery, the researchers synthesized a variety of lactone molecules through enzymatic C-H bond activation through hemiacetal intermediates.
Molecular dynamics simulations and quantum chemical calculations implied that the difference in product distribution between hemiacetals and lactones can be explained by the different binding preferences of cyclic ethers and hemiacetals toward the active site of the enzyme.