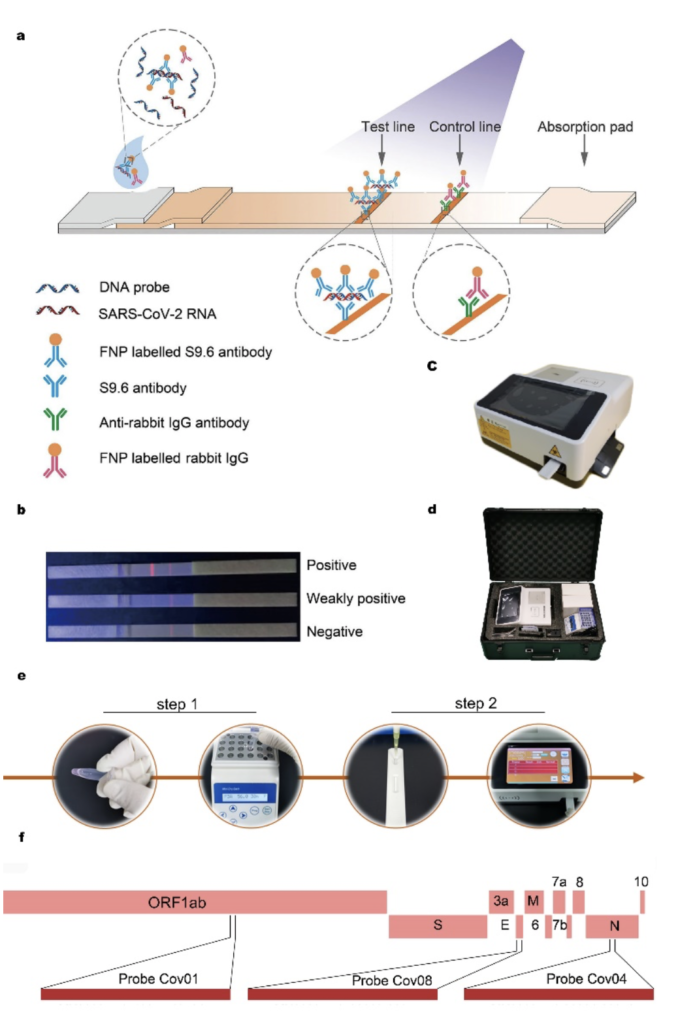
The hybrid-capture/FIA test is based on a monoclonal antibody S9.6 which recognizes DNA-RNA double-stranded hybrids from COVID-19 RNA coding for the conserved ORF 1ab, envelope protein and the nucleocapsid regions. S9.6-monoclonal-antibody-labelled europium-chelate-based fluorescent nanoparticles (FNPs) are then used to capture the hybridized double strands formed on a lateral flow strip. Hybridization time is 30 min, readout time for a fluorescent signal is 15 min.
The test kit requires little in terms of equipment and professional personnel and can function as a “suitcase laboratory” and to be used as an on-site detection method for outpatient and emergency departments, e. g., for customs and grassroots disease control. The test kit was already approved by the National Medical Products Administration and received European Conformity certification.
CAS news release, December 10, 2020
https://www.nature.com/articles/s41551-020-00655-z