http://english.cas.cn/newsroom/research-news/202604/t20260415_1156320.shtml
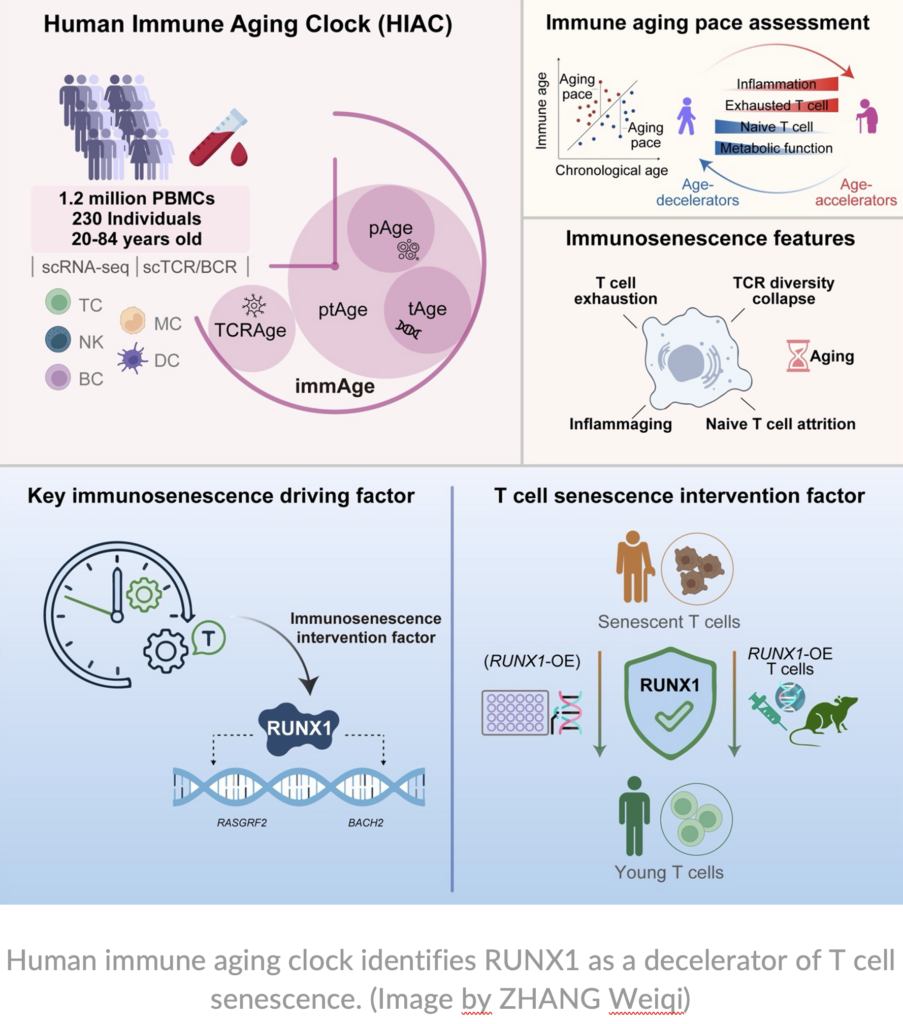
A research center affiliated with the Chinese Academy of Sciences has constructed a high-precision Human Immune Aging Clock (HIAC). The work systematically characterizes the multiscale dynamics of immunosenescence and identifies the transcription factor RUNX1 as a functional “brake” on T-cell senescence.
The team profiled peripheral blood samples from 230 healthy individuals spanning a 60-year age range, generating a high-resolution atlas of nearly 1.2 million peripheral blood mononuclear cells and identifying 24 immune cell subtypes.
The analysis revealed that aging induces profound remodeling of the immune landscape, marked by a sharp decline in naïve T cells and an expansion of exhausted T cells and of monocytes. This pattern reflects a state in which immune exhaustion coincides with chronic inflammation.
Individuals with decelerated immune aging displayed a more youthful immune profile, including higher proportions of naïve T cells, reduced senescence- and inflammation-associated transcriptional signatures, and plasma metabolomic profiles enriched with immunomodulatory and antioxidant molecules. These individuals also exhibited more favorable physiological traits, such as improved glycemic control, better liver function, and enhanced cardiopulmonary capacity.
The study identified a critical inflection point in human immune aging, with a pronounced peak in cellular and transcriptomic remodeling occurring around age 40. This finding highlights midlife as a key transition period for accelerated immune aging and points to a potential window for preventive intervention.
Through gene regulatory network analysis, the transcription factor RUNX1 emerged as a central regulator, with its downstream target genes contributing strongly to the immune aging clock. RUNX1 acts as a “youth-associated factor” in T cells: maintaining its expression helps counteract senescence, and its overexpression can reverse aging-related phenotypes.