https://www.cas.cn/syky/202604/t20260402_5105733.shtml
https://doi.org/10.1016/j.cell.2026.03.034
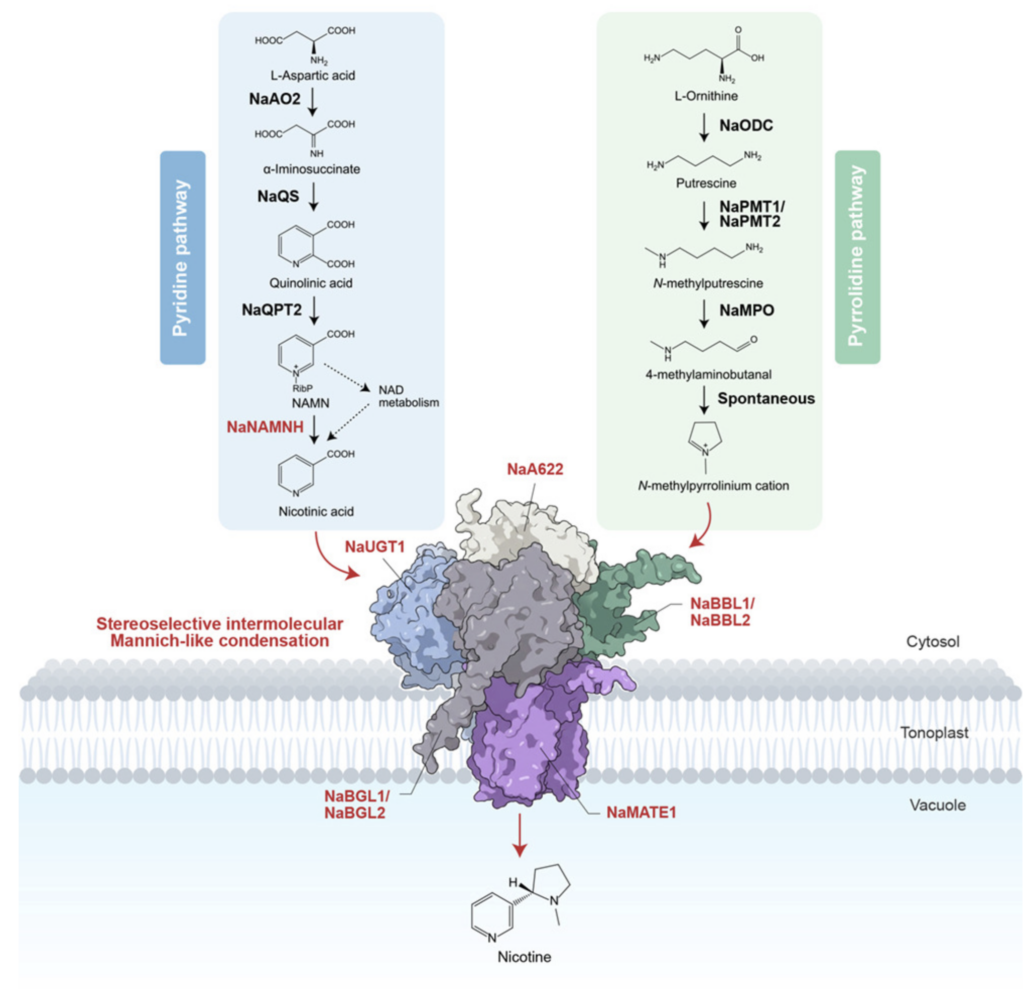
A team from the CAS Center for Excellence in Molecular Plant Sciences has elucidated the molecular mechanism of the intermolecular Mannich reaction in nicotine biosynthesis. The key finding was a nicotine-deficient mutant in wild tobacco which carried a key gene for nicotine pyridine ring synthesis.
The study found that the final synthesis of nicotine is synergistically achieved through a five-component dynamic metabolic pathway located in the vacuolar membrane. The entire process employs glycosylation and deglycosylation mechanisms. Nicotinic acid undergoes nitrogen atom glycosylation modification via glycosyltransferases to maintain its cationic state; subsequently, reductases reduce and decarboxylate it, activating the molecule; an intermolecular condensation with another pyrrolidine ring is completed through a stereoselective intermolecular Mannich reaction catalyzed by a berberine-like bridging enzyme, followed by subsequent oxidation; finally, deglycosylation by glucosyl hydrolases yields chiral, pure nicotine, which is stored in the vacuolar via the MATE transporter. This metabolic pathway achieves efficient transfer of intermediate products, avoids the accumulation of toxic intermediates, and prevents feedback inhibition of the synthetic pathway by high concentrations of products, thus solving the “autotoxicity dilemma” in plant self-defense.